Apps to track your prescriptions: Active Script List / My Script List
Many people now receive prescriptions as electronic “tokens” by SMS or email. This is convenient, but it can also become messy — especially if you have several regular medicines, repeats, or scripts from different doctors.
A useful option is the Active Script List, also known as My Script List or MySL. This is a digital list of your current active prescriptions, including electronic prescriptions and barcoded paper prescriptions. Once it is set up, new eligible prescriptions can be added automatically, and you do not need to hunt through old SMS messages for individual prescription tokens. (Australian Digital Health Agency)

How do you get started?
To use an Active Script List, you generally need to be registered through a participating pharmacy, or through a prescriber if their software supports it. In practice, for most patients, the easiest route is to ask their pharmacy to help. The Australian Digital Health Agency says patients can set up an ASL by visiting a pharmacy with a mobile phone and identification, and that most community pharmacies can register patients for the service. (Australian Digital Health Agency)
The pharmacy can provide an ASL activation code. This is the code you can then enter into a compatible app so the app can connect to your My Script List. MedAdvisor’s support information also states that patients must obtain an ASL activation code from a pharmacist and consent by SMS or email to link the account. (support.medadvisor.com.au)
You do not necessarily have to use the pharmacy’s own app
Many pharmacy chains have their own apps. These can be convenient, especially if you usually attend the same pharmacy.
However, you can also use more general prescription-wallet apps that are not tied to a particular pharmacy chain. Examples include:
- Scripty — https://www.getscripty.io
- MedAdvisor — commonly used by many pharmacies, but also supports Active Script List connection
Scripty says it can connect to ASL/MySL and automatically pull in new scripts and repeats once linked. Its app listing also describes automatic syncing with My Script List. (Scripty) MedAdvisor similarly provides instructions for connecting the app to Active Script List using an ASL activation code. (support.medadvisor.com.au)
Unlike the Pharmacy based appts, apart from the ASL activation code, Scripty does not require an 'Activation Code' from the Pharmacy. To obtain such you need to be on the Pharmacy's database ie you've been dispensed prescriptions or at least have provided your Medicare and other details. It is likely the same arrangement with MedAdvisor.
A practical example from the clinic
We recently helped a patient install Scripty on his phone while he was at the clinic. After installing the app, he logged in using his Google account, although other sign-in options were also offered.
He obtained his ASL activation code from a TerryWhite Chemmart pharmacy. This involved SMS consent. The pharmacy sent him a link to activate their own app, but we wanted to see whether the ASL code could also be used in a more generic app. It could: he entered the ASL code into Scripty and the app accepted it.
Interestingly, only one prescription appeared initially — a paper prescription. A recent eScript did not show up. One possible explanation is that the ASL may have been activated only after that eScript had already been dispensed, or that the prescription had not been added to the list in the expected way. This is an observation from one patient’s experience, not a definitive rule.
Important points
The Active Script List is optional. It requires your consent, and you remain in control of who can access your list. MySL describes the service as allowing patients and chosen health professionals to see prescriptions available for dispensing, with the patient controlling which pharmacies and doctors have access. (MySL)
It may also be possible to use an app without connecting ASL, but then the app may behave more like a manual script wallet rather than updating automatically. The main benefit of connecting ASL is that current scripts and repeats can sync automatically without relying on separate SMS tokens.
There is also a privacy and security consent process before these apps connect. Scripty is operated by Oexa Pty Ltd, and Scripty describes itself as integrating with My Script List / ASL. (Scripty) MySL’s terms explain that prescription information is accessed from prescription systems and displayed through authorised partners, subject to consent and the MySL framework. (MySL)
The take-home message
If you are comfortable using apps on your phone, the Active Script List can make prescription management easier. You can ask your pharmacy for an ASL / MySL activation code, then use either the pharmacy’s own app or a compatible general app such as Scripty or MedAdvisor.
You do not need to rely only on SMS prescription tokens, and you may not need to be locked into a single pharmacy chain’s app. The key is obtaining your ASL activation code and giving the required consent. Once linked, the app should be able to show your active prescriptions more conveniently in one place.





 Many Australians assume they won’t qualify for government concessions once they retire — particularly if they own assets or have super.
Many Australians assume they won’t qualify for government concessions once they retire — particularly if they own assets or have super.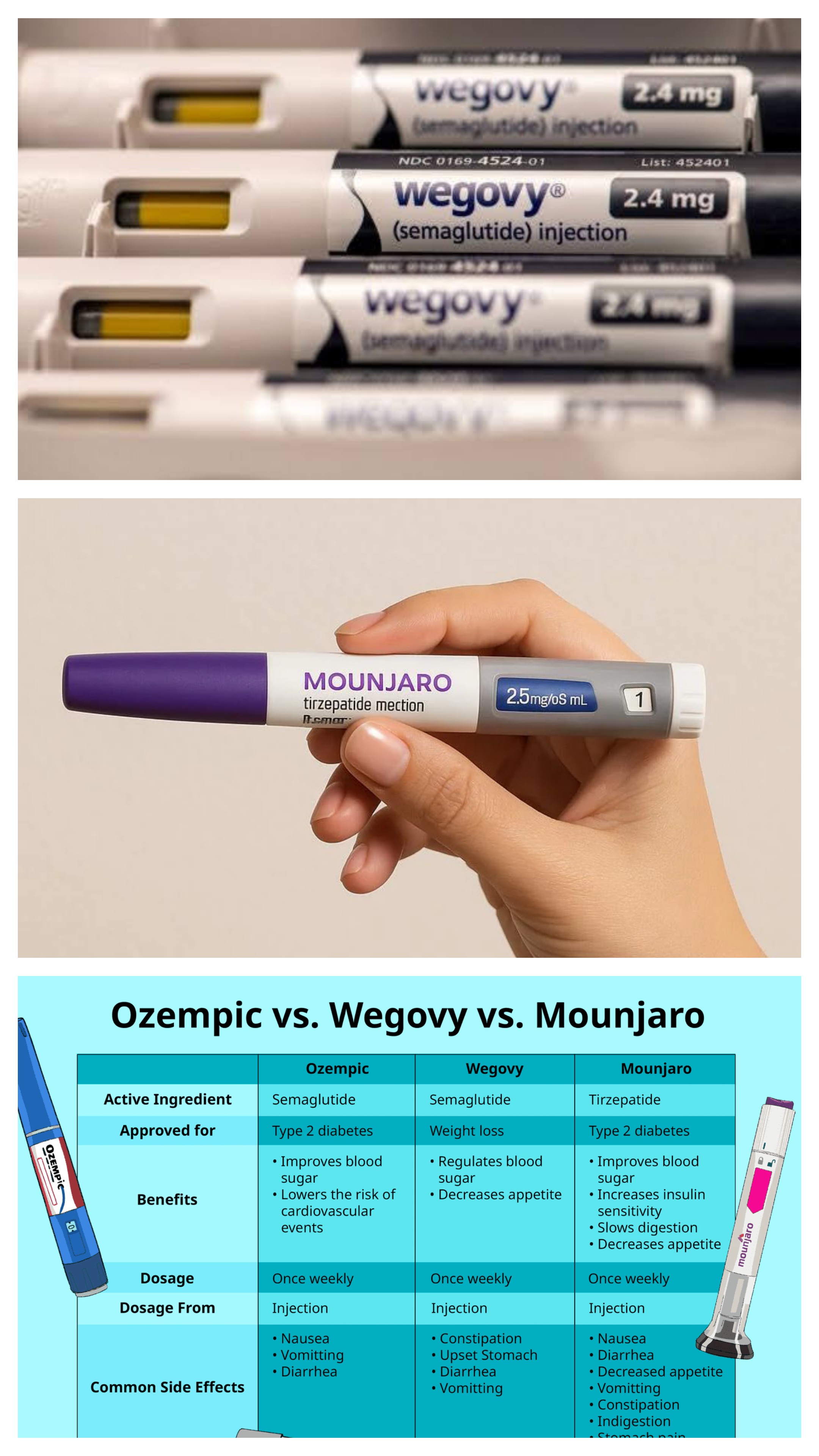 New medications known as GLP-1 receptor agonists are increasingly being used to assist with weight loss. These include:
New medications known as GLP-1 receptor agonists are increasingly being used to assist with weight loss. These include: